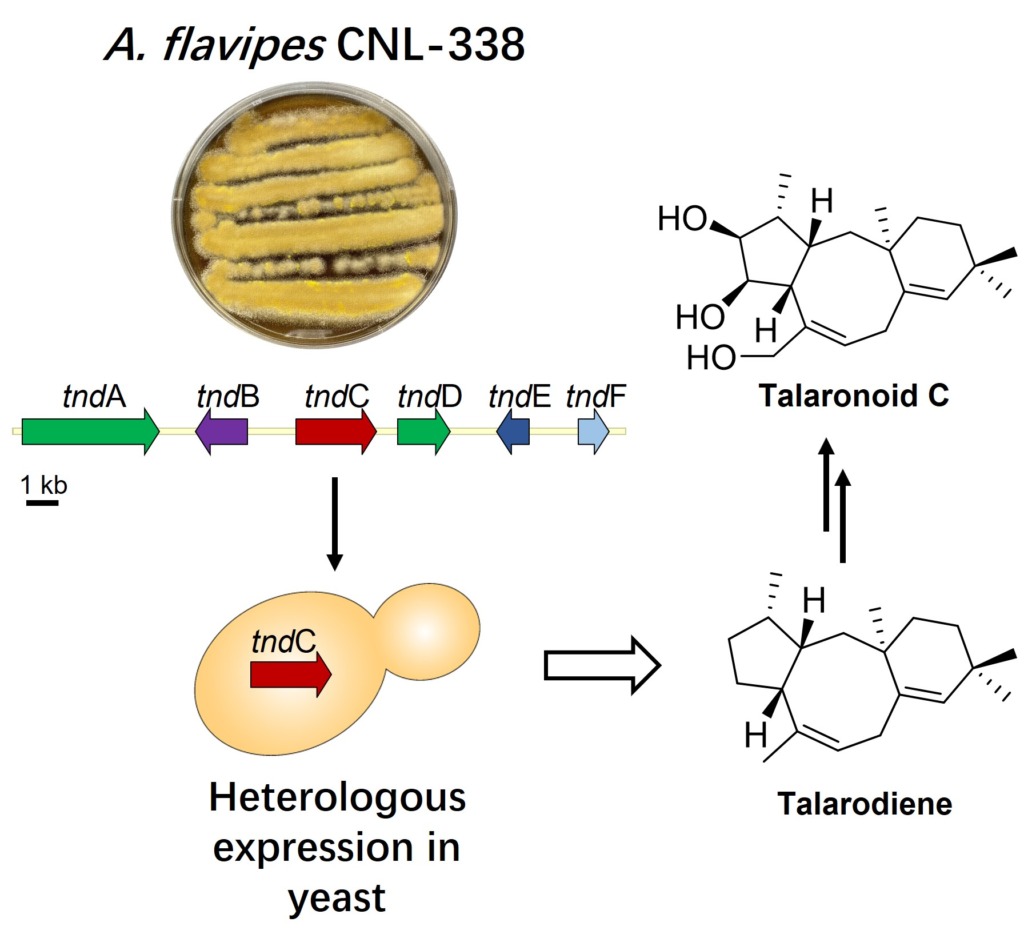
Discovery of new therapeutic agents from halophilic microorganisms isolated from Great Salt Lake
Environmental pressures have been shown to influence the chemical diversity of natural products and microorganisms isolated from extreme environments often produce molecules not observed in their terrestrial counterparts. The Great Salt Lake, also recognized as “America’s Dead Sea”, is an endorheic hypersaline lake located near the University of Utah. While seawater has an average salinity of ~3.3%, the Great Salt Lake ranges between 8-28%. Recently, our lab, in collaboration with Professor William Fenical’s group at Scripps Institution of Oceanography, started a natural products drug discovery campaign aimed at interrogating halophilic bacteria isolated from this unique environment.
Our preliminary data demonstrate that the unexplored hypersaline microorganisms of the Great Salt Lake produce metabolites containing molecular scaffolds never before observed, and their genomes contain unprecedented biosynthetic machinery. Thus, these microbes serve as an ideal resource for the discovery of natural products possessing novel scaffolds as well as the characterization of new biochemical reactions.
next